In the chemical equation Zn + 2HCL → ZnCl2 + H2, the reactants are A. zinc chloride and hydrogen. B. zinc - brainly.com
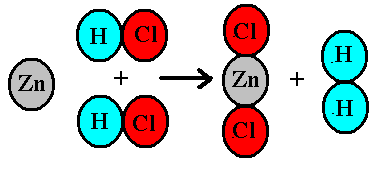
In the reaction Zn + 2HCl -> ZnCl_2 + H_2, how many moles of hydrogen will be formed when 4 moles of HCl are consumed? | Socratic

28. Columni Column II (p) 50% of excess reagent left (A) Zn(s) + 2HCl(aq) → ZnCl (s) + H2(g) above reaction is carried out by taking 2 moles each of Zn and